Worldwide demand for vaccine production has escalated exponentially since the vaccines for the ongoing coronavirus pandemic were developed. Multiple stakeholders including government and private organisations, NGOs, healthcare institutions, pharmaceutical companies, and more have come together to supply vaccines to the population. However, the global demand is extremely high and with the need for speedy and large-scale production and administration of vaccines, countries are facing the challenge of securely procuring, storing, and transporting the vaccine to thousands of healthcare facilities.
The two COVID-19 vaccines primarily being administered in India right now are Covishield and Covaxin, and both need to be taken in two doses. The time interval between the two doses varies for both vaccines. A third vaccine, Sputnik, has recently been introduced as well. The logistical complications of safely storing and distributing these vaccines are many, and a system that can identify each unique consignment supplied through various channels is the need of the hour.
Tracking vaccine movement accurately, creating end-to-end visibility at each stage of the supply chain and diligent record-keeping is important as it will help manufacturers and distributors keep track of their consignments in the supply chain. Keeping consistent records will be a great help to all key stakeholders in the vaccine supply chain, from distribution partners to healthcare workers.
To enable this, a standardised process to identify and track vaccines in the supply chain is required because every single consignment of vaccines changes many hands before reaching its final customer/patient, i.e., an individual to whom the dose will be administered. With each new stage of the supply chain, there is a new level of complexity that is added. In the past, vaccines have lost in transit, substandard product entered into the chain, or consignments reaching their destination with damaged or expired vials of the vaccine.
The present high vaccine demand and low supply offers a fertile ground for vaccine counterfeiters to try and make a quick profit. Several fakes have been identified, and even fake versions of drugs like Remdesivir have been found. Present circumstances are such that vaccines are not just prescribed to young children for illnesses dino tube that have already been eradicated – the coronavirus vaccines are one of the most important resources in the world right now. This makes the need for a clear supply chain that is traceable from the point of origin to the final stage in the process, an urgent and crucial one.
Pinay Viral || Asian Pinay || Pinay Kantutan || Pinay Scandal || Pinay Flix || Low Taper || Low Taper Fade || Dino Tube || Jujutsu Kaisen || Blowout Taper || Spicy Chat || Hydro Jug
Manufacturers or importers of vaccines should take the necessary steps to ensure that product labels carry standardised markings and utilise an end-to-end traceability system. Each record should also have key information like the batch number and expiry date, to enable validation of vaccines.
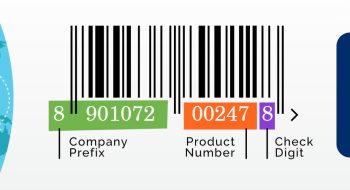
UNICEF has suggested a specification for a minimum basic traceability system to support countries avoid counterfeit or falsified medicines. Barcoding is vital in maintaining traceability, as each barcode is unique and can be identified as such. Unique identification encoded in barcodes is recommended by WHO on all levels of pinay flix vaccine packaging used for transit by manufacturers, except for the primary packaging of the product. A system that can lend ease and convenience to the tracking process, such as simply scanning a barcode, is very much required. It can facilitate efficient tracking of data and provides visibility in the supply chain so that recalling a consignment, for any reason, can be made easily.
GS1 India offers traceability service that meets global requirements, such as UNICEF standards, and has a host of benefits:
- It can help validate product barcodes, detect fraudulent consignments of vaccines and protect brand image and identity
- Provides accurate product information and control over product movement
- Batches in the supply chain can be efficiently tracked
- Helps reduce operational costs over an extended period of time
- Manual errors are eliminated and operations can run smoothly
- Provides better expiry management with First-In, First-Out (FIFO) system
- Identifies when stocks are low and need to be replenished
- Provides data insights to gain a better understanding of on-ground problems with the supply chain
Traceability, in the case of vaccines especially, is vital to ensure transparency across the supply chain and to ensure that the end consumers are safe. An end-to-end vaccine traceability helps perform recalls efficiently as and when required, from any stage of the supply chain.
Public Procurement’ to carry barcoding at the primary package level in 2019
With the production of vaccines constantly being accelerated to be able to keep up with global demands, traceability of vaccines and the capability for healthcare stakeholders to validate them at the point of administration is a pressing need. To enable this, each vaccine must be labelled with a 2D barcode and equipped with standardised information capture so as to fast-track logistical processes and track vaccines accurately no matter how far from the point of origin they have to be transported. Covert or forensic technologies can also be introduced on product packaging to add an extra layer of increased security to safeguard important resource from counterfeiters and fraudsters.